|
3/11/2023 0 Comments Bohr model of atom 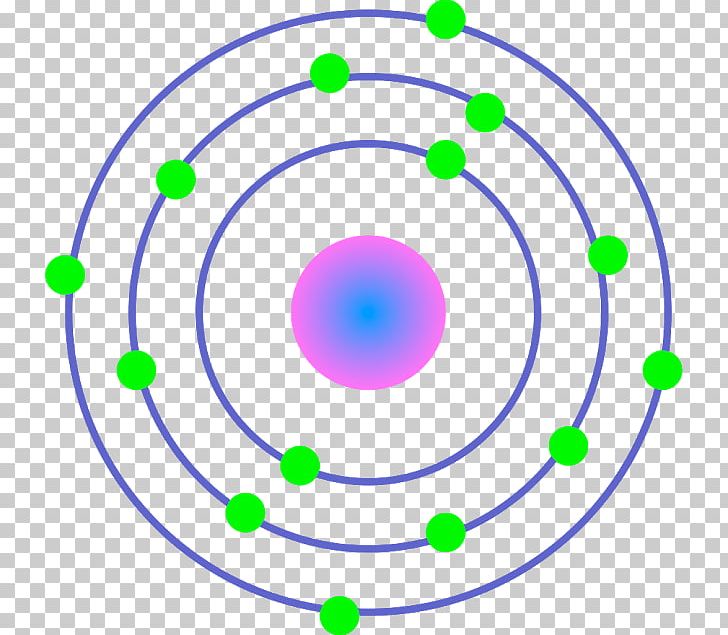
The Bohr model is used to account for the spectrum of the hydrogen atom, but the basic idea is the same for all elements. Light is only emitted at certain frequencies, each corresponding to a particular electronic transition within the allowed states. When this happens, the electrons lose some or all of the excess energy by emitting light. An excited atom is unstable and tends to rearrange itself to return to its lowest energy state. When an electron in an atom has absorbed energy it is said to be in an excited state. The lowest energy state an atom can be at is called its ground state. Each line in the spectrum corresponds to an electronic transition between energy levels associated with the element. Then the beam is sent through a glass prism to disperse the light into the spectrum. The spectrum is a result of applying some kind of energy, such as heat, to a sample and passing the light emitted through a slit, to obtain a narrow beam. This is what we call the Bohr model.Īs we all know, from our chemistry book, each element in the periodic table has its own characteristic emission spectrum. The Danish physicist that attempted to account for the emission spectrum of atomic hydrogen by proposing that the single electron in the hydrogen atom travels in definite circular orbits around the central nucleus and that only certain orbits were possible at certain energies. The spectrum of frequencies of radiation (light) emitted by an atom or a molecule in a transition from a higher state to a lower energy state.Ī condition or mode of being with regard to a set of circumstances position. An atom consists of a central positively charged nucleus surrounded by a sufficient number of negatively charged electrons to balance the nucleus. The smallest particle of an element, indivisible by chemical reaction, that can either exist alone or enter into chemical combination. A transition from a higher orbit to a lower orbit will release quantized energies of light, which would explain the light spectrum emitted by an element.Ī tentative description of a system or theory that accounts for all of its known properties. This is a model of the atomic structure in which electrons travel around the nucleus in well-defined orbits determined by quantum conditions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
